Explain the Difference Between Ionic Compounds and Covalently Bonded Compounds
Ionic bond full transfer of a valence electron. Ionic bonds are electrostatic in nature resulting from that attraction of positive and negative ions that result from the electron transfer process.

Chemistry For Kids Chemical Bonding Chemistry For Kids Covalent Bonding Chemical Bond
Drag the terms on the left to the appropriate blanks on the right to complete the sentences.

. As their names suggest ionic compounds are made of ionic bonds and covalent compounds are made of covalent bonds. An ionic bond essentially donates an electron to the other atom participating in the bond while electrons in a covalent bond are shared equally between the atoms. One of the atoms in the bond shall lose an electron to initiate the bond to form an ionic compound while the Covalent compound is formed by sharing the electrons among the atoms.
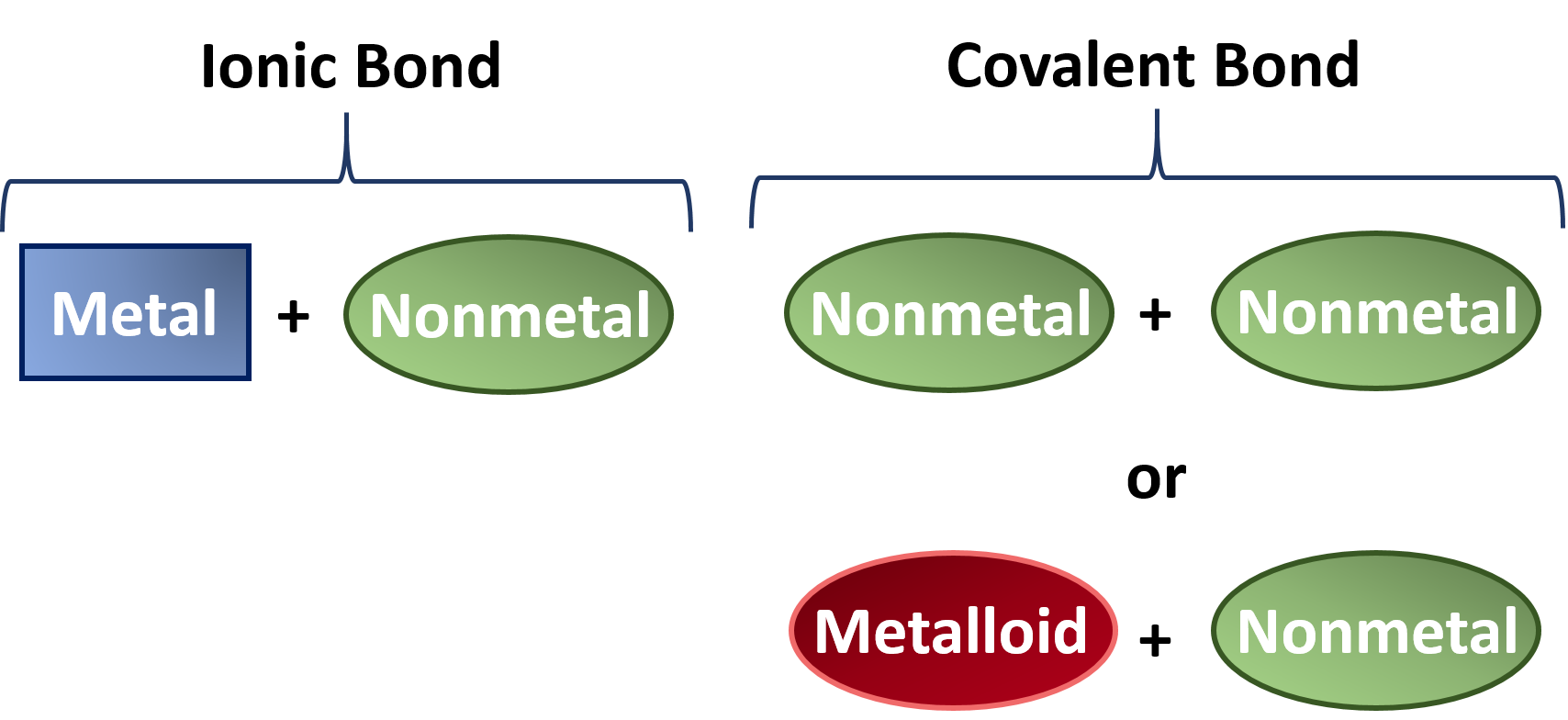
In general metallic elements tend to form ionic bonds and non-metallic elements tend to form covalent. The ionic bond is the electrostatic force of attraction between two oppositely charged ions. Covalent bond The covalent bond is also called a shared bond.
Ionic compounds tend to be hard and brittle while covalent compounds tend to be softer and more flexible. Electrons are shared in covalent bonds and electrons are exchanged in ionic bonds. Covalent and ionic compounds can be differentiated easily because of their different physical properties based on the nature of their bonding.
Covalent bonds are more prevalent than ionic bonds in Organic chemistry. When writing a covalent molecule use. Reset E metals and nonmetals 1.
At room temperature and normal atmospheric pressure covalent compounds may exist as a solid a liquid or a gas whereas ionic compounds exist only as solids. The main reason for these differences is the difference in their bonding pattern. Answer 1 of 90.
Prefixes Do not criss-cross when using. Explain the difference between an ionic bond and a covalent bond. Ionic bonds occur between two species which are electrostatically attracted towards each other whereas covalent atoms bond covalently through the sharing of electrons between their outer shells.
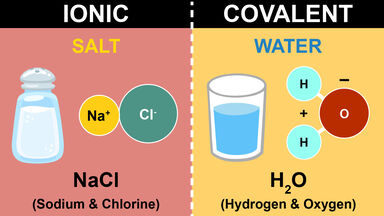
What is the difference between Covalent and Ionic Bonds. Which bond ionic or covalent involves cations and anions. Ionic bonds join metals to non-metals.
Metallic bonds are the chemical bonds that join metals to metals. Chem Ch 3-4 48 terms. Give an example of each.
When two atoms are close to each other they have a go at. Covalent bonds which occur between involve two or more nonmetals the. The primary difference between ionic and covalent bonds is that an ionic bond is a permanent transfer of valence electrons between two atoms.
Charge separation between covalently bonded atoms is. The rule is that when the electronegativity level is greater than 2 the bond is considered ionic. What is the difference between ionic and covalent compounds.
The chemical reaction between components in covalent bonds. Ionic bonds result from transfer of electrons whereas covalent bonds are formed by sharing. Ionic is losing and gaining electrons Covalent is sharing the same electrons Ionic - One atom loses and electron the other gains one and two oppositely charged ions are produced which are attracted to each other.
Lonic bonds which occur between involve the transfer of electrons from one atom to another 2. A covalent bond is formed from the mutual sharing of one or more pairs of electrons between two atoms non-metals. An ionic bond is formed as a result of the electrostatic attraction between the positive and negative ions formed by the transfer of electrons from one atomgroup to another.
Ionic bonds join metals to non-metals. The differences between each of the bonds is how strong the bond is between the atoms. Ionic compounds conduct electricity when dissolved in water while covalent compounds typically dont.
Lets dive right in. When the level is less than. Here are some differences.
Non-polar colavent bonding electrons are shared equally. At this point in education they have become scripture and often prevent understanding instead of helping it. What are Metallic bonds.
Covalent Bonds To show the sharing of electrons in a covalent bond you would. Additionally both bonds focus on the electrons. Circle the electrons being shared in each element.
On the other hand the covalent bond is sharing of electrons between two atoms. How do ionic bonds differ from covalent bonds when it comes to what happens to the electrons of the atoms involved in each type of bond. The ionic bond is the electrostatic force of attraction between two oppositely charged ions.
How does the formation of cationanions in the bond. Ionic compounds tend to have higher melting and boiling points than covalent compounds. Many differences can be noted between ionic and covalent compounds based on their macroscopic properties such as solubility in water electrical conductivity melting points and boiling points.
Therefore their bonding pattern can be deemed as the key difference between ionic and. These bonds join non-metals to non-metals. In this video we discuss how chemical bonds are formed we cover ionic bonds and covalent bonds.
A covalent bond is formed by the mutual sharing of electrons whereas an ionic bond is formed as a result transfer of electrons. Examples of ionic bonds are sodium chloride magnesium chloride magnesium oxide etc. Ionic and covalent bonds are the major two types of chemical bonds that exist in compounds.
Covalent - A shared pair of electrons resulting in both atoms having full outer shells. Ionic transfers electrons and covalent shares electrons. Polar colavent bonding electrons shared unequally.
Before going into the deep discussion let me give you a brief review of the two in a tabular form. Explain the differences between ionic and covalent bonds by answering the following questions. The only pure covalent bonds occur between identical atoms.
Click card to see definition -In an ionic bond the atoms are bound together by the attraction between oppositely-charged ions -In a covalent bond the atoms are bound by shared electrons Click again to see term 115 THIS SET IS OFTEN IN FOLDERS WITH. What are 5 properties of ionic bonding. One shares and the other trades.
Chemical bonding is important in many different functions o. Among living things there are more molecules formed by covalent bonding and it is common. The main difference between the Ionic and Covalent Compounds is the methodology of formation.
However by doing this both bonds create elements that are neutrally charged making them stable compounds. Ionic and covalent bonds are a man-made classifications to make understanding how atoms interact more easily. Ionic and covalent bonds are fundamentally different in the way they are formed.
Key Differences of Ionic vs Covalent. Explain the difference between ionic and covalent bonding. The main difference between covalent and ionic bonds is that ionic bonds occur between two species which are electrostatically attracted towards each other whereas covalent bonds occur covalently through the sharing of electrons between their outer shells.
The difference between ionic and covalent bond is that ionic bonds occur between atoms having very different electronegativities whereas covalent bonds occur between atoms with similar or very low electronegativity differences. This is the main difference between Ionic.

Ionic Vs Covalent Which Is Which And How To Tell Them Apart
Main Differences Between Ionic And Covalent Bonds

Science Homework Comic By Eiyecaieyre On Deviantart Chemistry Classroom Science Homework Science Chemistry

Ionic Bonds Vs Covalent Bonds Chemtalk

Solved 5 Differences Between Ionic Compound And Covalent Compound Brainly In
Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry

Comparison Between Covalent And Ionic Compounds Introduction To Chemistry
Difference Between Covalent And Ionic Bonds

Lesson Plan Bundle Ionic And Covalent Bonding Distance Learning Covalent Bonding Teaching Chemistry Molecular Geometry

Covalent Bonding Scaffolded Notes Covalent Bonding Ionic And Covalent Bonds Electron Affinity

Ionic And Covalent Bonding Ionic Compoundscovalent Compounds Transfer Electronsshare Electrons Can Form Single Double Or Triple Bonds Form Ions That Ppt Download
What Are The Uses Of An Ionic Compound Quora
Difference Between Ionic And Molecular Compounds

Difference Between Ionic And Covalent Bonds Compare The Difference Between Similar Terms

What S The Difference Between Ionic And Covalent Bonds
What S The Difference Between An Ionic Bond And A Covalent Bond Quora

Four Covalent Bonds Carbon Has Four Valence Electrons And Here A Valence Of Four Each Hydrogen Atom Has One Vale Covalent Bonding Chemical Bond Ionic Bonding

Chapter 6 2 Ionic And Covalent Bonding Ppt Video Online Download

Covalent Bonds Chemwiki Covalent Bonding Chemical Bond Chemistry Textbook
Comments
Post a Comment